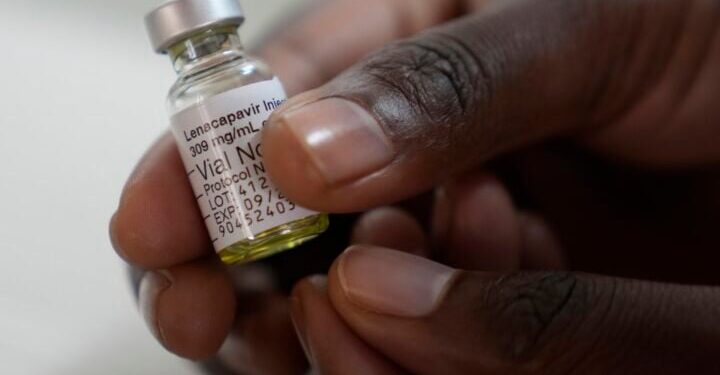
Nigeria is set to receive Lenacapavir, a long-acting HIV prevention drug, in March 2026.
The National Agency for the Control of AIDS (NACA) is advancing preparations for the introduction and rollout of Lenacapavir as pre-exposure prophylaxis (PrEP).
This injectable treatment is taken only twice a year and has shown significant reduction in HIV infection risk in clinical trials.
NACA has achieved regulatory approval from the National Agency for Food and Drug Administration and Control (NAFDAC) and completed landscape and readiness assessments in 10 states.
The agency is also conducting training for healthcare workers and developing information, education, and communication materials to support awareness creation.